Chlamydia spp.
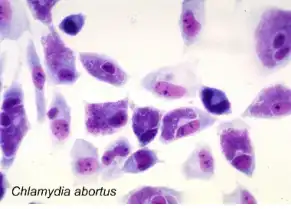
Mouse fibroblast cells infected with Chlamydia abortus. Purple-stained cytoplasmic inclusions containing chlamydial elementary bodies are visible in most cells.
Samples:
Notes: Send all samples at room temperature, or preserved in sample buffer.
Interpretation of PCR Results:
Chlamydia spp.
Chlamydia species are a group of obligately intracellular bacteria that replicate inside of a cytoplasmic inclusion. They form spore-like structures that are called elementary bodies and are released from host cells and infect other cells (Storz & Kaltenboeck in Woldehiwet_Chlamydial Dis 363, 1993). Chlamydiae are found in virtually all vertebrate and invertebrate animals. Currently nine pathogenic Chlamydia spp. are known that infect mammals.
Clinical Signs
Chlamydial diseases are typically not caused by toxins produced by the bacteria but by an ineffective cellular immune response that fails to eliminate the bacteria (Kaltenboeck Proc. 11th Int Symp Hum Chlamydial Inf 399, 2006). These diseases are typically chronic in nature, and manifest themselves as granulomatous lesions on mucosal membranes. Scarring lesions may be the consequence of repeated infections. Specific Chlamydia spp. are typically associated with specific hosts and disease manifestations (Storz & Kaltenboeck in Woldehiwet_Chlamydial Dis 363, 1993). Acute chlamydial diseases may present as respiratory diseases (C. pneumoniae, C. psittaci, C. pecorum, C. suis), enteritis, polyarthritis (C. pecorum), or urogenital infections (C. trachomatis, C. suis, C. pecorum) The main chlamydial diseases of companion animals are feline respiratory infections caused by C. felis, and avian respiratory, intestinal, and generalized infections caused by C. psittaci. Both pathogens may also infect pet owners and may cause conjunctivitis (C. felis) or mild to severe or even fatal respiratory infection (C. psittaci).
Standard Diagnostic Methods
The variability of signs makes clinical diagnosis difficult. An additional difficulty in culture isolation is the low amount of the organisms present in chronic lesions, and the need for cell culture or chicken embryos to propagate the bacteria. Various PCR methods are used for detection of chlamydial DNA or RNA, and ELISA, CFT and microimmunofluorescence methods are used for retrospective serological diagnosis (Kaltenboeck Proc. 11th Int Symp Hum Chlamydial Inf 399, 2006).
Our Method
The quantitative PCR approach we have developed targets the highly conserved 23S ribosomal RNA gene of Chlamydia spp. (DeGraves BioTechniques 34_106, 2003; Jee JCM 42_5664, 2004). This method, used with duplex fluorescent oligonucleotide probes, detects single genome copies of all known Chlamydia spp. and differentiates the major groups among the 9 species (C. psittaci, C. abortus, C. felis, C. caviae – C. pecorum – C. pneumonia – C. trachomatis – C. suis – C. muridarum).
